 Modeling the Latent Heat of Fusion
Modeling the Latent Heat of Fusion
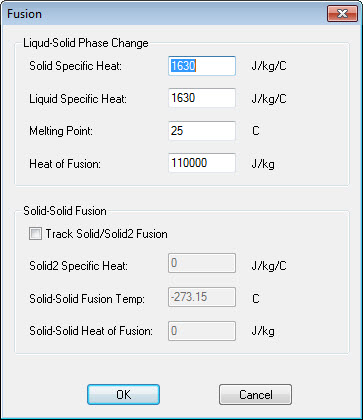
Tracking the energy of a phase change can be difficult if your software isn't designed to handle it. One trick of yesteryear was to create a spike in the thermal capacitance versus temperature table, but this is not particularly accurate and can cause problems with convergence. Thermal Desktop® provides a user friendly interface for defining the thermophysical properties of a phase changing materials, and SINDA/FLUINT accurately models this energy. This interface allows for a liquid/solid phase change as well as an additional solid/solid.
This can be applied to something as simple as melting ice, but specific phase-change materials (PCMs) are designed to be used across many industries such as solar thermal storage, solar power plants, medical devices, electronics, spacecraft, and food storage. The intent is to absorb or release significant amounts of energy while maintaining a constant temperature.
A sample model was created to show how to set up a phase change material. A transmitter is mounted to a convectively cooled aluminum plate. The transmitter is in a low power mode most of the time, but at fixed intervals it goes into a high power mode to transmit. A PCM was applied to the aluminum plate to dampen the thermal response of the transmitter.

This example is available on our User Forum
See also this example of a wax canister in a three-way mixing valve
